Holy SHIT. Pfizer & the FDA were FORCED to release this data by a federal judge. The list of deadly adverse reactions of note is frankly breathtaking. They KNEW.
— Shannon Joy (@ShannonJoyRadio) March 1, 2022
Pfizer data submitted to FDA contains 8 pages of known side effects. Just a sampling herehttps://t.co/oZ2dEFVX7Z… pic.twitter.com/3qJAiO3p0s
This document seems to contain information up to March of 2021.
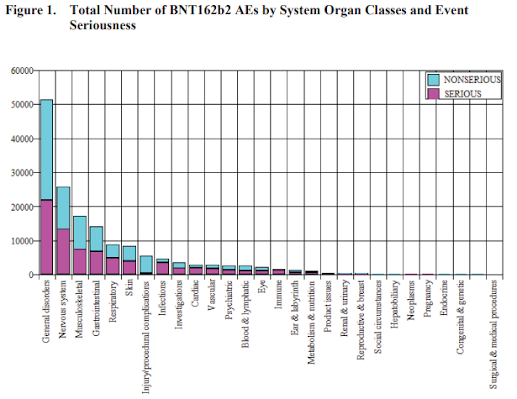
METHODOLOGY Pfizer is responsible for the management post-authorization safety data on behalf of the MAH BioNTech according to the Pharmacovigilance Agreement in place. Data from BioNTech are included in the report when applicable. Pfizer’s safety database contains cases of AEs reported spontaneously to Pfizer, cases reported by the health authorities, cases published in the medical literature, cases from Pfizer-sponsored marketing programs, non-interventional studies, and cases of serious AEs reported from clinical studies regardless of causality assessment.
... Due to the large numbers of spontaneous adverse event reports received for the product, the MAH has prioritised the processing of serious cases, in order to meet expedited regulatory reporting timelines and ensure these reports are available for signal detection and evaluation activity.
The increased volume of reports has not impacted case processing for serious reports, and compliance metrics continue to be monitored weekly with prompt action taken as needed to maintain compliance with expedited reporting obligations. Non-serious cases are entered into the safety database no later than 4 calendar days from receipt.
Entrance into the database includes the coding of all adverse events; this allow for a manual review of events being received but may not include immediate case processing to completion. Non-serious cases are processed as soon as possible and no later than 90 days from receipt.
Pfizer has also taken a multiple actions to help alleviate the large increase of adverse event reports. This includes significant technology enhancements, and process and workflow solutions, as well as increasing the number of data entry and case processing colleagues.
To date, Pfizer has onboarded approximately additional fulltime employees (FTEs). More are joining each month with an expected total of more than additional resources by the end of June 2021.
... Search criteria: Standard Decreased Therapeutic Response Search AND PTs Dyspnoea; Tachypnoea; Hypoxia; COVID 19 pneumonia; Respiratory Failure; Acute Respiratory Distress Syndrome; Cardiac Failure; Cardiogenic shock; Acute myocardial infarction; Arrhythmia; Myocarditis; Vomiting; Diarrhoea; Abdominal pain; Jaundice; Acute hepatic failure; Deep vein thrombosis; Pulmonary embolism; Peripheral Ischaemia; Vasculitis; Shock; Acute kidney injury; Renal failure; Altered state of consciousness; Seizure; Encephalopathy; Meningitis; Cerebrovascular accident; Thrombocytopenia; Disseminated intravascular coagulation; Chillblains; Erythema multiforme; Multiple organ dysfunction syndrome; Multisystem inflammatory syndrome in children.
Pfizer performs frequent and rigorous signal detection on BNT162b2 cases. The findings of these signal detection analyses are consistent with the known safety profile of the vaccine. This cumulative analysis to support the Biologics License Application for BNT162b2, is an integrated analysis of post-authorization safety data, from U.S. and foreign experience, focused on Important Identified Risks, Important Potential Risks, and areas of Important Missing Information identified in the Pharmacovigilance Plan, as well as adverse events of special interest and vaccine administration errors (whether or not associated with an adverse event). The data do not reveal any novel safety concerns or risks requiring label changes and support a favorable benefit risk profile of to the BNT162b2 vaccine.
No comments:
Post a Comment